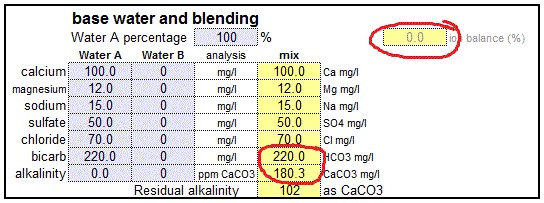
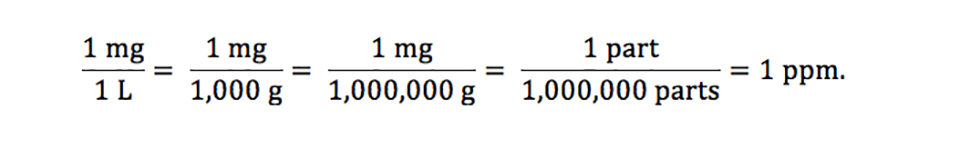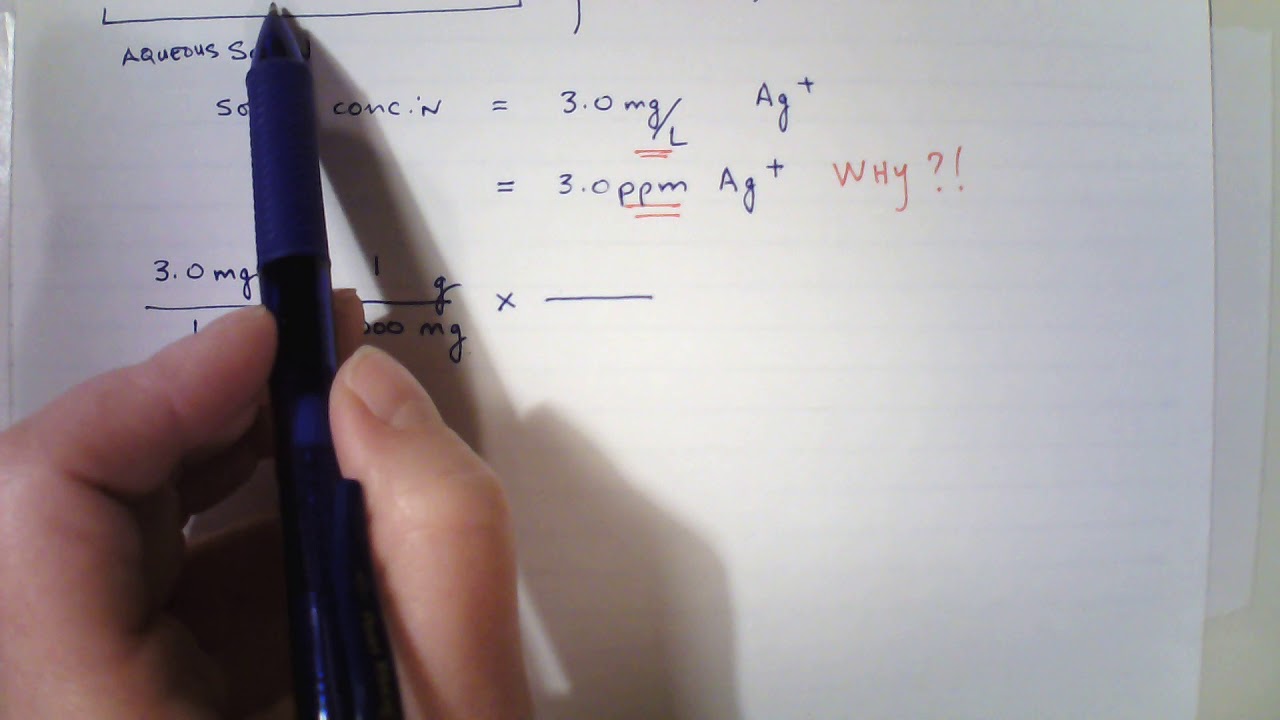SOLVED: 6. Calculate the concentration of Mn2+ in ppm (mg/L) in Mn solution of 0.21 M (Mn 54.9 glmol): (1 Point) 12500 ppm 1055 ppm 987 ppm 11500 ppm

Pounds Formula Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit. - ppt download

Concentrations expressed as mg L -1 (ppm) by labs must be divided by... | Download Scientific Diagram